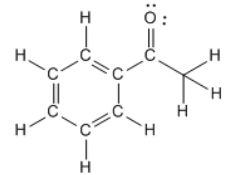
The molecular formula explains a given molecule’s number and types of atoms. We need to understand their molecular and structural formulas to count sigma and pi bonds within organic molecules. Calculating Sigma and Pi Bonds in Organic Molecules: Similarly, triple bonds consist of one sigma bond plus two pi bonds, each located above and below its counterpart. A double bond forms when two orbitals overlap directly, creating one sigma bond any remaining electrons form two pi bonds – one above and one below it – above and below this initial sigma bond. Relationship Between Sigma and Pi Bonds:ĭouble and triple bonds consist of one sigma or one or two pi bonds, depending on the form. To identify pi bonds within a molecule, look for multiple bonds like double or triple links their number equals that number. Pi bonds typically appear in molecules with double or triple bonds, such as C=C or CC. That creates an electron density concentration above and below this axis. Understanding Pi Bonds:Ī pi bond is a covalent bond formed when two atomic orbitals overlap, parallel to the bond axis. The number of sigma bonds depends on how many atoms are directly bonded to one another within that molecule. 
To identify them in a molecule, look for single bonds or those not involved in aromaticity. Examples include C-H, C-C, and C-N bonds. In simpler terms, it’s an arrangement in which the electron density is concentrated between them. 
A sigma bond is a covalent bond formed when two atoms directly overlap along their bond axis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
